Cochlear Implants Linked to New Bone Formation, Increased Hearing Loss
Released: December 07, 2021
At A Glance
- New bone formation that can adversely affect long-term hearing preservation was seen in cochlear implant recipients.
- New bone formation that can adversely affect long-term hearing preservation was seen in cochlear implant recipients.
- Of 123 patients, 83 (68%) had new bone formation, predominantly located at the base of the cochlea, within four years of implantation.
- RSNA Media Relations
1-630-590-7762
media@rsna.org - Linda Brooks
1-630-590-7738
lbrooks@rsna.org - Imani Harris
1-630-481-1009
iharris@rsna.org
Cochlear implantation is a well-established means to treat severe hearing loss. The device consists of an external part that sits behind the ear and a second part surgically implanted under the skin that stimulates nerves in the cochlea, a fluid-filled, spiral structure in the inner ear that transmits sound across sensory nerves to the brain. Cochlear implants don't restore normal hearing, but they can help a person recognize words and better understand speech.
While complications from cochlear implants are rare, post-mortem studies have linked them with inflammation, fibrosis and new bone formation. The potential clinical implications of new bone formation make it desirable to visualize and possibly prevent development, but in-vivo—or within the body—detection has not yet been described.
"Such subtle changes are challenging to visualize in vivo, in particular in the vicinity of a metallic implant causing artifacts on computed tomography images," said study co-lead author Floris Heutink, M.D., M.Sc., from the Department of Otorhinolaryngology at Radboud University Medical Center (Radboudumc) in Nijmegen, the Netherlands. "However, through the new availability of an ultra-high-resolution CT scanner, we were encouraged to investigate this."
Dr. Heutink and colleagues used ultra-high spatial resolution CT (UHRCT) to assess new bone formation and its implications in 123 patients with cochlear implants.
The researchers were able to detect new bone formation in vivo using UHRCT. Of the 123 patients, 83 (68%) had new bone formation, predominantly located at the base of the cochlea, within four years of implantation. Long-term residual hearing loss was significantly larger in the group with new bone formation.
"As indicated by our study, there is a correlation between new bone formation and long-term residual hearing loss," said study co-author Berit M. Verbist, M.D., Ph.D., from the Departments of Radiology at Radboudumc and Leiden University Medical Center in Leiden, the Netherlands.
New bone formation around the cochlear implant electrode has a number of negative consequences due to its effects on the device and the structures surrounding it. It can affect the spread of electrical current within the cochlea, leading to complex device fitting, channel interaction and poorer overall hearing outcome. It could also complicate future therapies such as gene therapy to restore cochlear function.
"Last but not least, new bone formation may complicate reimplantation surgery," Dr. Verbist said.
The researchers noted that improvements in design and surgical techniques have made cochlear implantation available for an increasing number of people with severe to profound hearing loss. This increase in patients makes a method for detecting and monitoring new bone formation in vivo more urgent than ever. Still, many remaining questions have to be answered, Dr. Heutink said, before the detection of new bone formation in an individual patient would have therapeutic consequences.
"This technique will be a valuable tool to gain insight into occurrence, time course and the pathophysiology of this process and maybe used to evaluate still-to-be-developed treatments against new bone formation," he said.
"Ultra-High-Resolution CT to Detect Intracochlear New Bone Formation after Cochlear Implantation." Collaborating with Drs. Heutink and Verbist were Tim M. Klabbers, M.D., Wendy J. Huinck, Ph.D., Federica Lucev, M.D., Willem Jan van der Woude, B.Sc., and Emmanuel A. M. Mylanus, M.D., Ph.D.
Radiology is edited by David A. Bluemke, M.D., Ph.D., University of Wisconsin School of Medicine and Public Health, Madison, Wisconsin, and owned and published by the Radiological Society of North America, Inc. (https://pubs.rsna.org/journal/radiology)
RSNA is an association of radiologists, radiation oncologists, medical physicists and related scientists promoting excellence in patient care and health care delivery through education, research and technologic innovation. The Society is based in Oak Brook, Illinois. (RSNA.org)
For patient-friendly information on CT, visit RadiologyInfo.org.
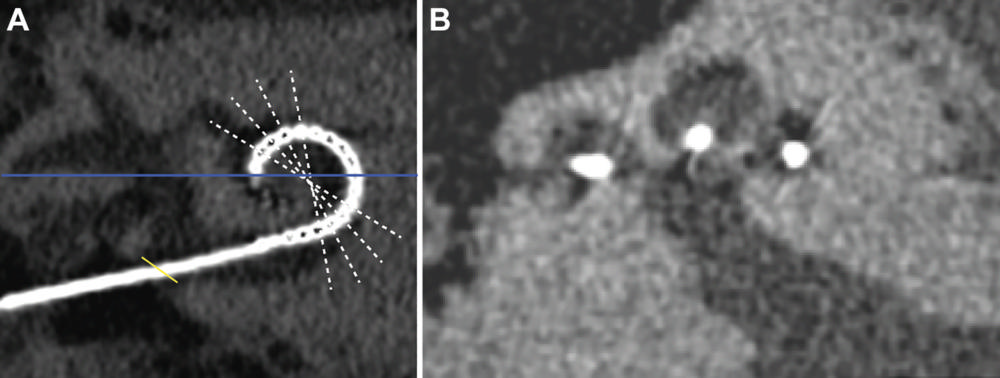
Figure 1. Methods for CT assessment. (A) An oblique multiplanar reconstruction through the basal turn of the cochlea was obtained and (B) midmodiolar sections were obtained with radial multiplanar reconstruction through the center of the cochlea (white lines in A). On such midmodiolar images, the presence or absence of new bone formation and the scalar position were assessed at each electrode contact. Angular insertion depth was measured from the lateral border of the horizontal semicircular canal (blue line in A), using a correction factor to the center of the round window (yellow line in A) of 34°.
High-res (TIF) version
(Right-click and Save As)
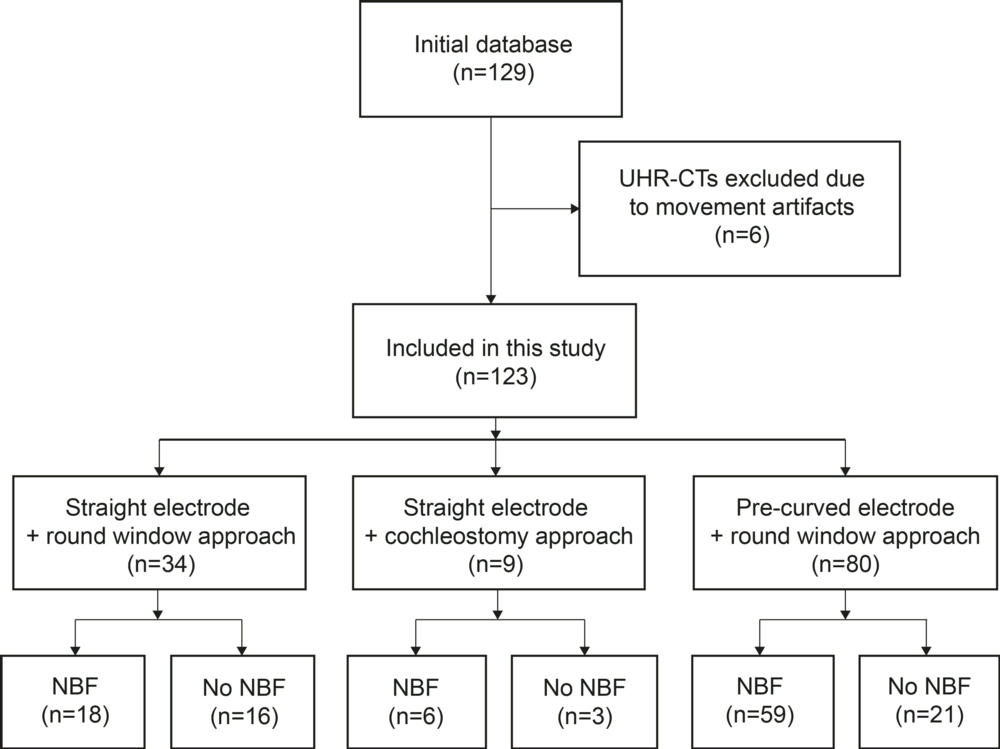
Figure 2. Flowchart of included cochlear implant recipients with ultra-high-spatial-resolution (UHR) CT for evaluation of intracochlear new bone formation (NBF).
High-res (TIF) version
(Right-click and Save As)
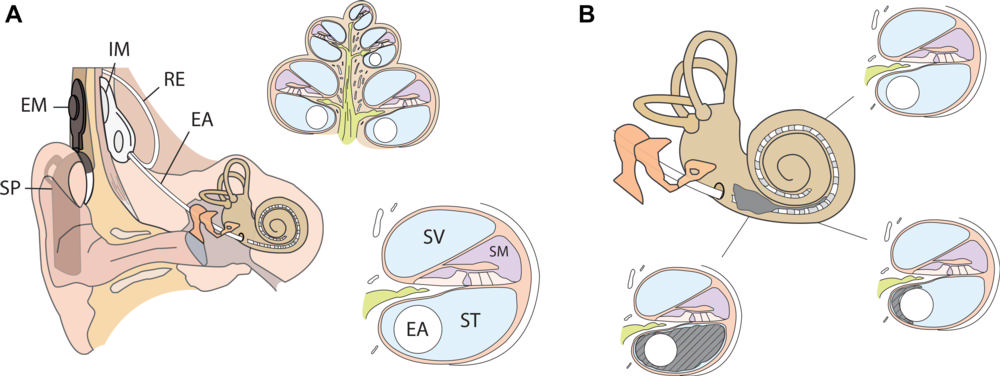
Figure 3. Schematics of (A) right ear with cochlear implant, midmodiolar cross-section, and close-up of the different compartments within the cochlea and (B) proximal new bone formation (in gray) in the scala tympani (ST) encasing the electrode array (EA), gradually diminishing toward the apex of the cochlea. EM = external magnet, IM = internal magnet, RE = reference electrode, SM = scala media, SP = speech processor, SV = scala vestibuli.
High-res (TIF) version
(Right-click and Save As)
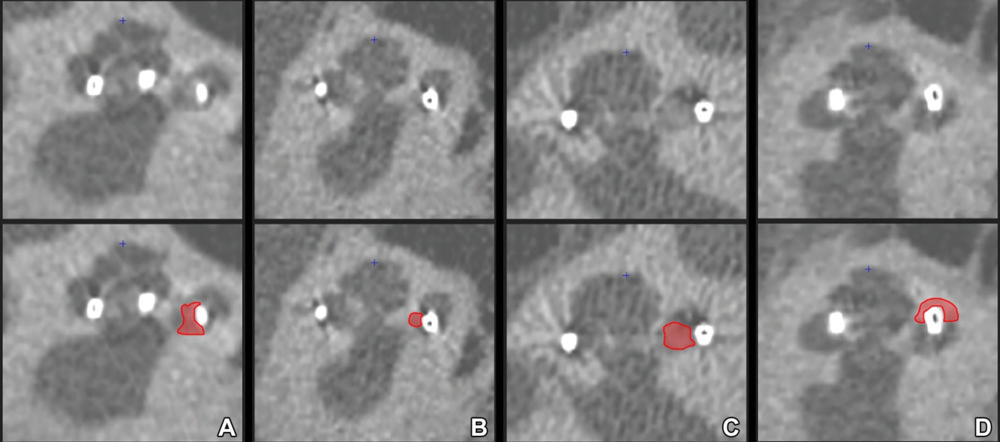
Figure 4. Midmodiolar ultra-high-spatial-resolution CT scans with corresponding annotations indicating new bone formation (NBF) in red. Note the varying appearance of NBF, ranging from (A) slightly increased attenuation, resembling that of the modiolus, to (D) frank ossification with an attenuation similar to that of the otic capsule surrounding a translocated contact in the scala vestibuli. An intermediate form of NBF appearance is shown in B and C. Moreover, in B, the ossification between the perimodiolar positioned electrode contact and the medial cochlear wall is difficult to discern, resulting in interrater disagreement.
High-res (TIF) version
(Right-click and Save As)
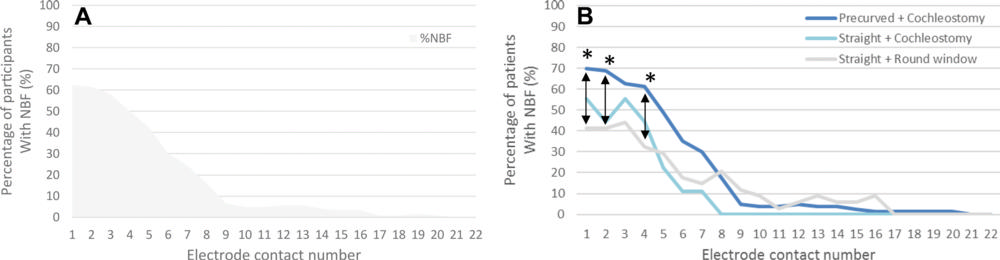
Figure 5. Graphs show percentage of participants with new bone formation (NBF) per electrode contact location (A) for all participants and (B) according to the three surgical combinations. * = P < .05.
High-res (TIF) version
(Right-click and Save As)
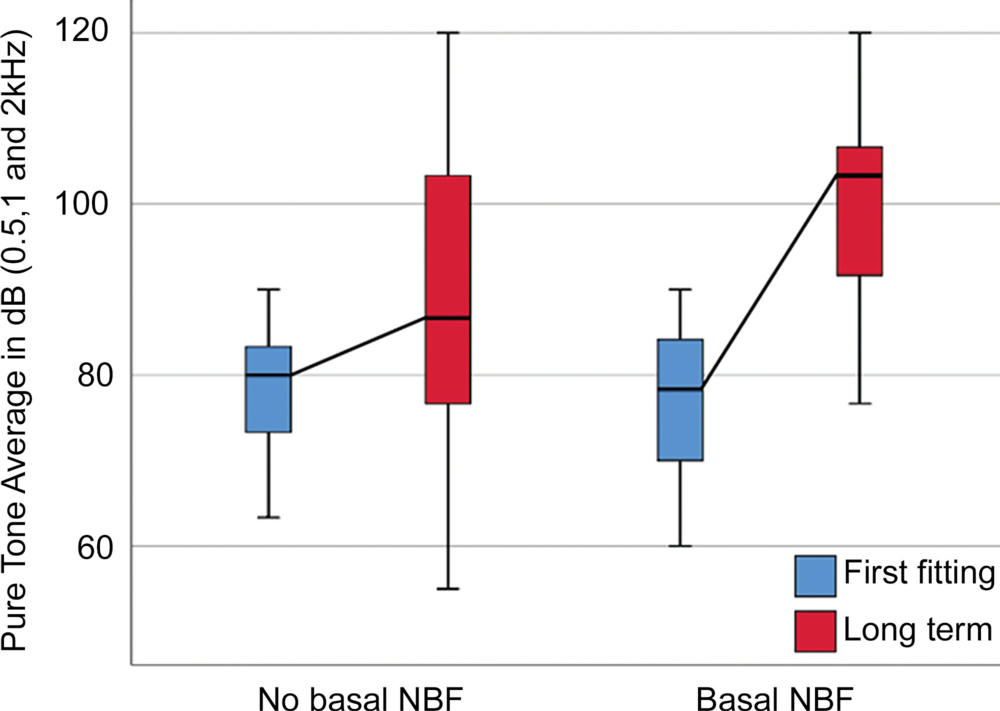
Figure 6. Box plot compares residual acoustic hearing loss in the ear with cochlear implant between the first fitting (the initial activation and calibration of the cochlear implant 4–6 weeks after surgery) and long-term follow-up for participants with and without new bone formation (NBF).
High-res (TIF) version
(Right-click and Save As)
