Low-dose Chest CT Leaves DNA Intact
Released: March 10, 2020
At A Glance
- Lung cancer screening with low-dose chest CT does not appear to cause damage to human DNA.
- Researchers compared the DNA in 107 patients who underwent low-dose chest CT with that of 102 who had standard-dose chest CT.
- Analysis of the DNA found significant differences between the group that had a standard-dose chest CT scan and those who had a low-dose chest CT.
- RSNA Media Relations
1-630-590-7762
media@rsna.org - Linda Brooks
1-630-590-7738
lbrooks@rsna.org - Dionna Arnold
1-630-590-7791
darnold@rsna.org
OAK BROOK, Ill. — The low-dose chest CT scans used in lung cancer screening do not appear to damage human DNA, according to a study appearing in the journal Radiology. The results could help allay fears that such screenings will lead to an increase in radiation-induced cancer.
Low-dose CT screening for lung cancer in high-risk patients such as longtime smokers gained favor after the National Lung Screening Trial. The trial reported that use of low-dose chest CT scans could significantly reduce deaths from lung cancer compared to screening with chest X-rays. CT was able to identify cancers at an earlier, more treatable stage.
Along with the promise of CT screening came worries over the effects of radiation exposure on patients, as even the low-dose exam delivers more radiation than an X-ray—radiation that could affect DNA and potentially lead to cancer. Studies of these potential effects that rely on epidemiology, or the analysis of diseases in the population at large, have limitations, according to study senior author Satoshi Tashiro, M.D., Ph.D., director of the Research Institute for Radiation Biology and Medicine at Hiroshima University in Hiroshima, Japan. A biological approach that looks at the effects of exposure on DNA has more power, he said.
“The National Lung Screening Trial suggested the value of low-dose CT screening in high-risk population for developing lung cancer,” he said. “There were, however, no studies investigating the biological effect of low-dose CT scans on large numbers of patients. These findings led us to investigate these effects.”
Dr. Tashiro and colleagues developed a system to look for damage and abnormalities in chromosomes, strands of DNA wound into a double helix structure inside the cell. In a previous study, the technology showed increases in chromosomal aberrations after standard CT scans.
For the new study, the researchers compared the DNA in 107 patients who underwent low-dose chest CT with that of 102 who had standard-dose chest CT. They obtained blood samples before and 15 minutes after CT. The median effective radiation dose of low-dose CT was 1.5 millisieverts (mSv). The standard CT dose was 5.0 mSv.
Analysis of the DNA found significant differences between the group that had a standard-dose chest CT scan and those who had a low-dose chest CT.
“We could clearly detect the increase of DNA damage and chromosome aberrations after standard chest CT,” Dr. Tashiro said. “In contrast, no significant differences were observed in these biological effects before and after low-dose CT.”
Although low-dose CT is now commonly used for screening exams, standard CT is an effective diagnostic tool that is appropriate when the benefits outweigh any potential risk.
While the study did not endorse lung cancer screening with low-dose CT, its results appear to ease concerns over a potential increase in radiation-related cancer risk related to screening programs.
“Even using these sensitive analyses, we could not detect the biological effects of low-dose CT scans,” Dr. Tashiro said. “This suggests that application of low-dose CT for lung cancer screening is justified from a biological point of view.”
Beyond lung cancer screening, Dr. Tashiro said the DNA analysis could be used to study the biological effects of other types of imaging.
“We are interested in the biological effects of various types of radiological diagnosis, including PET/CT, to establish a better system for the management of medical radiation exposure,” he said.
“Biological Effects of Low-Dose Chest CT on Chromosomal DNA.” Collaborating with Dr. Tashiro were Hiroaki Sakane, M.D., Mari Ishida, M.D., Ph.D., Lin Shi, Ph.D., Wataru Fukumoto, M.D., Ph.D., Chiemi Sakai, Ph.D., Yoshihiro Miyata, M.D., Ph.D., Takafumi Ishida, M.D., Ph.D., Tomoyuki Akita, Ph.D., Morihito Okada, M.D., Ph.D., and Kazuo Awai, M.D., Ph.D.
Radiology is edited by David A. Bluemke, M.D., Ph.D., University of Wisconsin School of Medicine and Public Health, Madison, Wisconsin, and owned and published by the Radiological Society of North America, Inc. (https://pubs.rsna.org/journal/radiology)
RSNA is an association of radiologists, radiation oncologists, medical physicists and related scientists promoting excellence in patient care and health care delivery through education, research and technologic innovation. The Society is based in Oak Brook, Illinois. (RSNA.org)
For patient-friendly information on CT, visit RadiologyInfo.org.
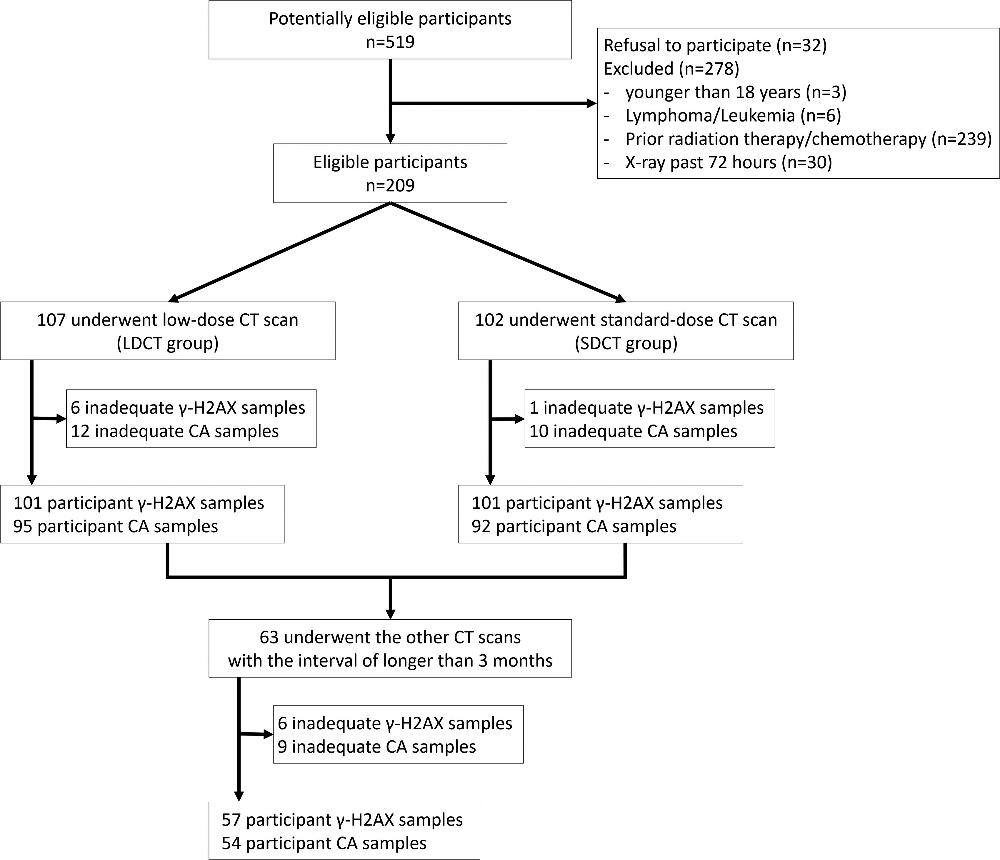
Figure 1. Study participant enrollment flowchart. γ-H2AX, a marker of DNA double-strand break, is a phosphorylated type of histone γ-H2AX. CA = chromosome aberration, LDCT = low-dose CT, SDCT = standard-dose CT.
High-res (TIF) version
(Right-click and Save As)

Figure 2. Immunofluorescent staining performed to depict γ-H2AX foci. Representative images of γ-H2AX foci in peripheral blood lymphocytes in an 82-year-old woman who underwent standard-dose CT. (a) Nuclear DNA of four lymphocytes. (b) γ-H2AX foci (arrows). (c) Markers of DNA double-strand breaks. In this merged image, DNA is blue and γ-H2AX foci are red (arrows show small foci). γ-H2AX, a marker of DNA double-strand breaks, is a phosphorylated type of histone H2AX. Scale bar: 5 mm.
High-res (TIF) version
(Right-click and Save As)

Figure 3. Peptide nucleic acid fluorescence in situ hybridization analysis performed to depict chromosome aberrations. Metaphase lymphocyte in an 87-year-old man obtained 15 minutes after standard-dose CT. Representative images of dicentric chromosomes with two centromeres (arrow) and of acentric fragments (arrowhead) are shown. The acentric fragments were created by the formation of dicentric chromosomes. (a) Chromosomal DNA. (b) Telomeres. (c) Centromeres. (d) Merged image. Blue = DNA, green = telomeres, red = centromeres.
High-res (TIF) version
(Right-click and Save As)

Figure 4. Box and whisker plot shows the absolute number of γ-H2AX foci before and after CT in two independent groups. The number of γ-H2AX foci was significantly increased after standard-dose CT (SDCT). Before and after low-dose CT scanning there was no significant difference (LDCT). ⁎ Wilcoxon signed-rank test was applied. † Mann-Whitney U-test was applied. Adjusted P values are shown after Bonferroni correction. γ-H2AX, a marker of DNA double-strand break, is a phosphorylated type of histone H2AX.
High-res (TIF) version
(Right-click and Save As)
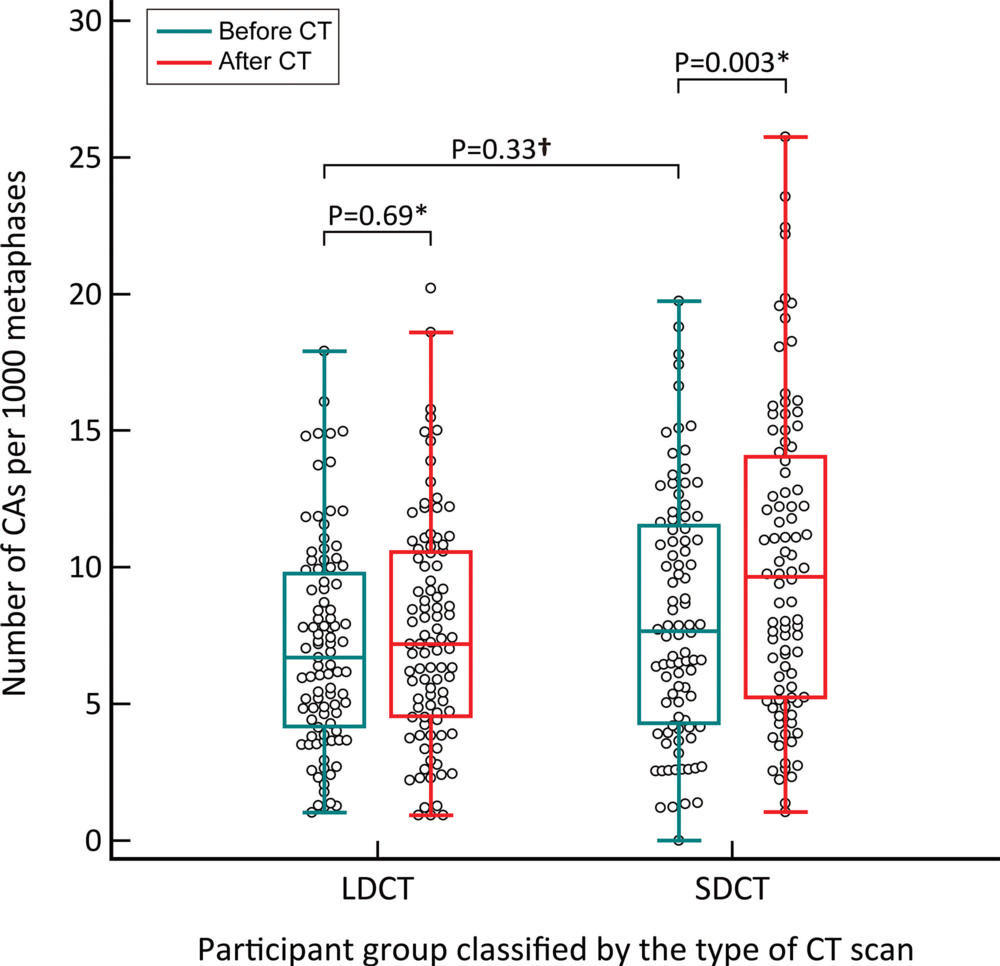
Figure 5. Box and whisker plot show the absolute number of chromosome aberrations (CAs) before and after CT in two independent groups. The number of CAs was significantly higher after standard-dose CT (SDCT). There was no significant difference before and after low-dose CT (LDCT group). ⁎ Wilcoxon signed-rank test was applied. † Mann-Whitney U-test was applied. Adjusted P values are shown after Bonferroni correction.
High-res (TIF) version
(Right-click and Save As)
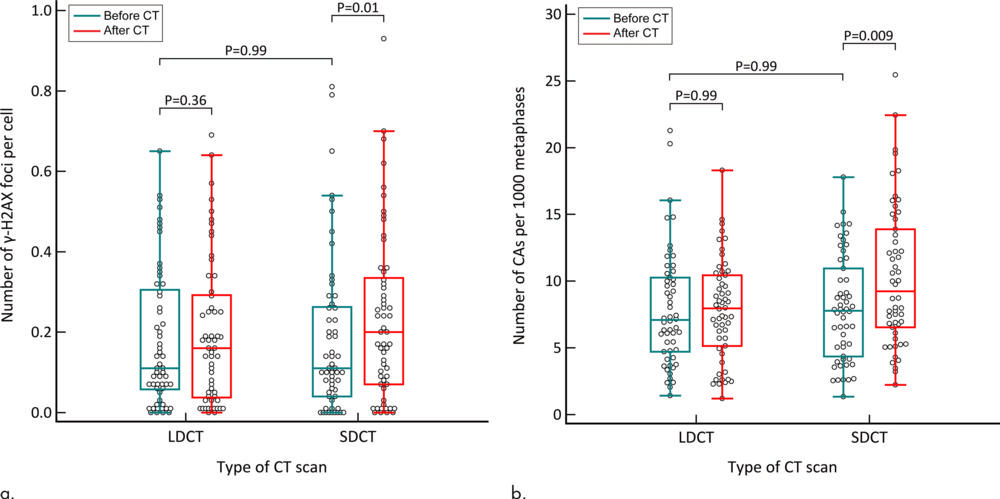
Figure 6. Box and whisker plots show the absolute number of DNA damages in the participants who underwent both low-dose CT (LDCT) and standard-dose CT (SDCT). (a) γ-H2AX foci analysis. (b) Chromosome aberration (CA) analysis. The number of both γ-H2AX foci and CAs was significantly increased after standard-dose CT. There was no significant difference before and after low-dose CT. The Wilcoxon signed-rank test was applied. Adjusted P values are shown after Bonferroni correction. γ-H2AX, a marker of DNA double-strand break, is a phosphorylated type of histone H2AX.
High-res (TIF) version
(Right-click and Save As)
