Study Suggests Intracranial Pathology Not Necessary for Gadolinium Deposition in Brain Tissues
Released: June 27, 2017
At A Glance
- Traces of gadolinium are retained in the brain years after MRI.
- Intracranial gadolinium deposition appears to occur among patients with a normal, intact blood brain barrier.
- Approximately 400 million patients have undergone gadolinium-enhanced MRI.
- RSNA Media Relations
1-630-590-7762
media@rsna.org - Linda Brooks
1-630-590-7738
lbrooks@rsna.org
OAK BROOK, Ill. Minute traces of gadolinium, the contrast agent used to enhance MR images, are often retained in the brain tissue of patients years after undergoing MRI. Some studies have suggested this deposition was limited to patients with underlying brain pathology, such as a tumor or infection. However, new research suggests gadolinium retention may be more widespread and may be present in many more, or possibly all, patients exposed to gadolinium-based contrast agents, according to new research published online in the journal Radiology.
Gadolinium contrast media were introduced as a means of enhancing MRI images in 1988 and are commonly used today. The contrast agent is injected into the patient’s bloodstream and eliminated from the body through the kidneys.
“It’s estimated that approximately 400 million doses of gadolinium have been administered since 1988,” said the study’s lead researcher, Robert J. McDonald, M.D., Ph.D., staff neuroradiologist at the Mayo Clinic in Rochester, Minn. “Gadolinium contrast material is used in 40 to 50 percent of MRI scans performed today.”
Scientists previously believed gadolinium contrast material could not cross intact blood brain barrier, the semipermeable membrane that selectively filters materials from the bloodstream from entering extracellular fluid in the brain and central nervous system.
“By late 2014, the first evidence emerged that gadolinium was depositing within brain tissues. However, many of these patients had underlying medical conditions, such as brain tumors, that could adversely affect the blood brain barrier,” Dr. McDonald said. “We were curious if this deposition might be related to the integrity of the blood brain barrier, so we studied patients with normal brain pathology and presumably an intact blood brain barrier.”
The study involved an analysis of post-mortem neuronal tissue samples from five patients who had undergone four or more (up to 18) gadolinium-enhanced MRI exams between 2005 and 2015 and 10 patients who had undergone MRI exams without gadolinium. The median age at the time of death was 68 years in the contrast agent-exposed group and 79 years in the control group. All patients in the current study were exposed to gadodiamide (Omniscan).
During the patient autopsies, a small amount of brain tissue was obtained from each patient and studied with transmission electron microscopy, mass spectroscopy and X-ray spectroscopy. The analysis revealed dose-dependent gadolinium deposits in four neuroanatomical regions of the brain in patients who underwent contrast-enhanced MRI. At the time of autopsy, all study patients had normal brain pathology.
“Our results suggest current thinking with regard to the permeability of the blood brain barrier is greatly oversimplified, as gadolinium appears to accumulate even among patients with normal brain tissue and no history of intracranial pathology.” Dr. McDonald said. “It will take additional research to understand how and why this deposition is occurring.”
The analysis revealed that certain areas of the brain, including the globus pallidus and dentate nucleus, retained more of the gadolinium agent.
“These neuroanatomical regions of the brain are also areas prone to mineralization and hemorrhage, which may suggest that parts of the brain may have a less robust barrier and may be more susceptible to this deposition,” Dr. McDonald said.
Because free gadolinium is similar to calcium in size and charge, Dr. McDonald said the body may also mistake the contrast media for this endogenous metal, which is often taken up in areas of the brain during the aging process.
“These are all questions that need to be further explored,” he noted.
Despite finding evidence of gadolinium contrast agent deposition in the neural tissues, the researchers detected no histologic changes to suggest toxicity. The FDA has identified no convincing scientific evidence to date that these deposits are harmful, but is further investigating this matter.
“Gadolinium-based contrast agents have revolutionized MRI and diagnostic medicine,” Dr. McDonald said. “They have allowed us to detect diseases that would otherwise be extremely difficult to diagnose.”
“Gadolinium Deposition in Human Brain Tissues after Contrast-enhanced MR Imaging in Adult Patients without Intracranial Abnormalities.” Collaborating with Dr. McDonald were Jennifer S. McDonald, Ph.D., David F. Kallmes, M.D., Mark E. Jentoft, M.D., Michael A. Paolini, M.D., David L. Murray, M.D., Ph.D., Eric E. Williamson, M.D., and Laurence J. Eckel, M.D.
Radiology is edited by Herbert Y. Kressel, M.D., Harvard Medical School, Boston, Mass., and owned and published by the Radiological Society of North America, Inc. (http://radiology.rsna.org/)
RSNA is an association of over 54,600 radiologists, radiation oncologists, medical physicists and related scientists, promoting excellence in patient care and health care delivery through education, research and technologic innovation. The Society is based in Oak Brook, Ill. (RSNA.org)
For patient-friendly information on MRI, visit RadiologyInfo.org.
Images (.JPG and .TIF format)
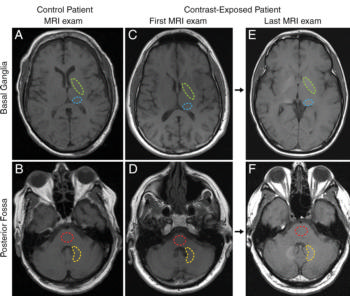
Figure 1. Graphs show gadolinium detection with mass spectrometry of cadaveric tissues. A–D, Changes in gadolinium ion signal intensity detected with mass spectrometry plotted against cumulative intravenous gadolinium exposure for each neuroanatomic area. Strength of association between gadolinium ion signal intensity and dose is shown with Spearman rank correlation coefficient (r) and associated P value.
High-res (TIF) version
(Right-click and Save As)
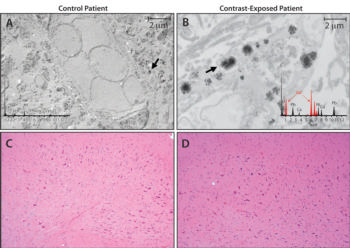
Figure 2. Tissue localization and cellular response to gadolinium deposition. A, B, Micrographs from transmission electron microscopy (0.2 percent lead citrate stain; original magnification, × 10,000) show cellular localization of gadolinium in dentate nuclei tissue samples from, A, control patient 7 and, B, gadolinium-exposed patient 1. X-ray spectra are shown in inset of each respective panel for selected electron-dense foci (arrows); gadolinium peaks in spectra are indicated by red overlay. C = carbon, Ca = calcium, Cs = cesium, Cu = copper, Gd = gadolinium, Ni = nickel, O = oxygen, Os = osmium, Pb = lead, Ti = titanium, V = vanadium. C, D, Photomicrographs from light microscopy (hematoxylineosin stain; original magnification, ×100) of dentate nuclei samples from, C, control patient 7 and, D, gadolinium-exposed patient 1.
High-res (TIF) version
(Right-click and Save As)
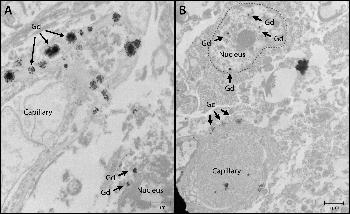
Figure 3. Nuclear localization of gadolinium (Gd) deposits. Images from transmission electron microscopy (0.2 percent lead citrate stain) show cellular localization of gadolinium in dentate nuclei tissue samples from, A, gadolinium-exposed patient 1 and, B, gadolinium-exposed patient 5 at 2,000–10,000-fold magnification. X-ray spectra were collected for selected electron-dense foci (arrows) to verify their identity.
High-res (TIF) version
(Right-click and Save As)
